|
7/28/2023 0 Comments Atomic size trend 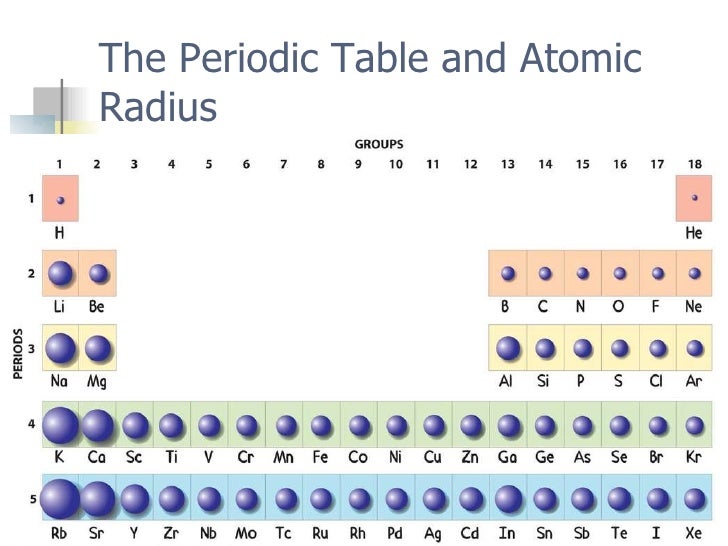
Consider, for, example, the internuclear distance between two hydrogen atoms in the H2 molecule is known to be 74 pm. The units for measuring the atomic radii are picometers, which is equal to 10−12 meters. Let us learn how the atomic radius is measured and what is its unit.Ītomic radii are measured for elements. There are three types of atomic radii which are Van der Waals radius, covalent radius and ionic radius. However, since the boundary is not well-defined, there are several non-equivalent definitions of the atomic radius. The atomic radius of an element refers to the measure of the size of the element’s atoms, which is typically the mean distance from the nucleus centre to the boundary of its surrounding shells of the electrons. Today, we will learn about what is atomic radius periodic table, the atomic radius definition, what is the atomic radius trend, what is atomic size periodic table, and the atomic size trend. We can, therefore, define the atomic radius of elements as half of the distance between the nuclei of similar atoms which are bonded together. For standardizing the measurement of the atomic radius, the distance between the nuclei of two similar atoms that are bonded together is measured. However, these orbital boundaries are quite fuzzy and tend to vary in different conditions. The atomic size is defined by its orbital edge. It helps us in understanding why some of the molecules fit in together and the rest contain parts which get crowded under different conditions. One of the ways in which we can express the size of an atom is with the help of atomic radius. While we learn about the behaviour of a compound or an atom, the size of the atoms plays an essential role. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
